
IT Tools
A suite of integrated and customizable IT tools, developed in-house, to optimize the management of your clinical projects and the collection of reliable and consistent data
IT TOOLS,
SEAMLESS CONNECTIVITY
AND INTUITIVE DESIGN
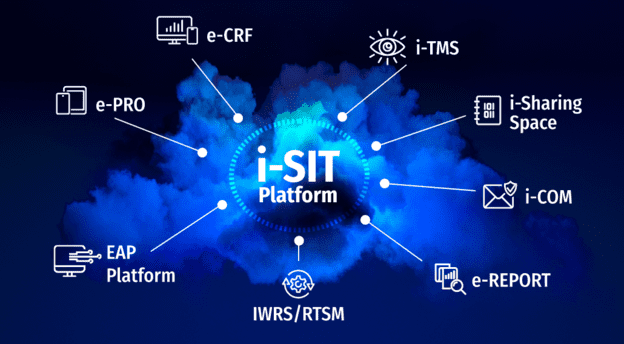
The i-SIT platform
The i-SIT platform is based on a single, unified data repository, shared by all ICTA IT tools.
For each study, data is stored once and made available to all departments and software that need to use it. This ensures data consistency, as there is no longer a need to manage silos and reconcile data from multiple sources.
All this contributes to increased confidence in the data within the organisation, which has a direct impact on operational performance.
ICTA WEB-BASED INTEGRATED SUITE OF TOOLS
i-TMS
The clinical operations hub to simplify study conduct and improve visibility
Our CTMS is designed to provide intuitive tracking of information and logical organisation of data. i-TMS enables stakeholders to track and report on the progress of clinical projects, and to make informed decisions based on quality data.
- Fully integrated with SOPs and FORMs (forms can be adapated to the needs of the sponsor)
- Monitoring and online monitoring reports
- Integrated validation process
e-REPORT
Online progress reports to support decision-making
Sponsor has access to online progress reports with possibility to export data with sorting and filtering functionalities (Excel files, graphs, charts). These reports provide a snapshot of the study progress, thus facilitating oversight of the clinical study and stakeholders activities, and support decision-making.
e-CRF
Integrated IT and Data Management development for consistent data
The ICTA eCRF is user-friendly with thorough validation documentation for electronic data entry. It is entirely hosted on ICTA servers in France .
- Developed in-house in compliance with the most advanced quality standards
- Compatible with main browsers on PC, Mac and mobile devices
- Customised home page: curves, newsletters, study documentation, etc.
- Overview of data entry, queries, signed visits, etc.
- Secure access defined per profile (Investigators, CRAs, DM, Sponsors, etc.)
- Automatic notifications and alerts (new patient, SAE etc.)
- Source Data Verification (SDV) management including audit trail and query management
- Remote monitoring activity management
- Fully integrated with data management activities from data management plan to database lock
e-PRO
The direct link between the patient and the clinical study
The ICTA ePRO is a robust and reliable system for patient-reported outcomes. It addresses our clients’ needs, can be customized to suit the study speficities, and provides quality data.
Principles of native application
- Accessible on smartphones/tablets with responsive design
- Transfer of data
Conditions to access the application
- Inclusion of the patient by the investigator
- After randomisation, unique access code issued and sent to the patient by the investigator
- Code associated to a password defined by the patient
Development platforms
- Mobile platforms used: development of a web application supported by the main types of platforms: iOS, Android, Windows
IWRS/RTSM
Random and Trial Supply Management solutions
The system proposed by ICTA allows fine-tuning of the randomisation algorithms and supply rules developed by the ICTA randomisation manager.
- Randomization
- Unblinding
- Supply / Resupply
- Acknowledgement of Receipt
- Dispensation
i-COM
Secure portal for communication and exchange of documents with the sites
- Accessible on smartphones/tablets with responsive design
- Send/Answer/View of messages
–> From/To ICTA study team to site participants
–> From/To sponsor to site participants - Secured transmission of documents with limited patient identification data
- Global/grouped communication to sites (reminders)
EAP Platform
Secure and integrated data flows to address the specificities of Early Access/Compassionate Use programs
Functionalities
- Management and progress monitoring of EAP/CUP
- Tracking of administrative data on hospitals, HealthCare Providers (HCPs), and patients
- Follow-up of treatment orders
- Management of safety data
- Recording of contacts with HCPs to recover missing data and optimize consistent data collection
- Generation of standard EAP reports including a general report and detailed reports of included patients, refusals, treatment orders and contact details of all active HCPs
- Compliant with new requirements (Pasrel/ATIH and multi-factor authentication)
Electronic Data Capture
- Data entry by HCPs
- Online controls
